💥 Debunking the Lie: “Studies Show Opioids Don’t Work for Chronic Non-Cancer Pain”
🚫 Debunking the Space Trial Claim
Krebs, E. E., Gravely, A., Nugent, S., Jensen, A. C., DeRonne, B., Goldsmith, E. S., ... & Noorbaloochi, S. (2018).
Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: The SPACE randomized clinical trial.
JAMA, 319(9), 872–882.
https://doi.org/10.1001/jama.2018.0899
Free Printable Resources: Please subscribe (free) to our Patreon Page
⚡TL;DR - The Truth About the Space Trial
The 2018 SPACE Trial is often used to claim that “opioids don’t work” for chronic pain. But that’s a gross misrepresentation of what the study actually showed.
THE CLAIM:
The 2018 SPACE Trial “proved” that opioids are ineffective for chronic pain.
THE TRUTH:
The SPACE Trial showed both opioids and non-opioids worked — and neither group was superior for function. The trial excluded most chronic pain patients, used low opioid doses, and still found no cases of addiction, OUD, or death.
Despite this, the study was spun by media, regulators, and expert witnesses to justify policies that have devastated pain patients — including forced tapers, medication denial, and criminalization of care.
This wasn’t science. It was strategy.
📚Table of Contents
- Intro: Debunking the SPACE Trial Narrative
- What the SPACE Trial Actually Found
- How the Narrative Was Spun by Media and Advocacy Groups
- Who Benefited: Conflicts, Litigation & Paid Experts
- How It Was Misused to Harm Patients
- What the Study Really Means
✅Section 1: Debunking the SPACE Trial Narrative
In 2018, a study called the SPACE Trial was published in JAMA and quickly seized by media outlets, addiction advocates, and policymakers. They claimed it “proved” that opioids are ineffective for chronic pain, especially long-term. This single study was cited in headlines, courtrooms, federal guidelines, and insurance policy changes — despite being limited in scope, deeply misrepresented, and filled with nuances the public was never told about.
The result? Millions of people living with severe pain were denied treatment, tapered off stable doses, or forced to try medications that didn’t work — all because of how this one study was spun. The SPACE Trial became a blunt weapon in a war on pain care. This article sets the record straight — not just on what the study actually found, but on who benefited from its distortion, and why pain patients are still paying the price.
📊Section 2: What the SPACE Trial Actually Found
The 2018 Strategies for Prescribing Analgesics Comparative Effectiveness (SPACE) Trial was a randomized study of opioid vs. non-opioid medications in 240 U.S. veterans with chronic back pain or osteoarthritis (OA) of the hip or knee. It was published in JAMA and quickly weaponized to claim that “opioids don’t work for chronic pain.” But when you actually read the study, that’s not what it shows.
🧪 Study Design
- Conducted within the VA system (2013–2015)
- 12-month comparison of opioid vs. non-opioid therapy
- Patients had chronic back or OA pain (moderate/severe)
- All participants were opioid-naïve
- The trial used a “treat-to-target” approach — adjust meds to improve function
- Primary outcome: Pain-related function
- Main secondary outcome: Pain intensity
Who Was Excluded?
The trial excluded most of the chronic pain population, including:
- People with fibromyalgia, migraines, or other complex conditions
- Anyone already on chronic opioid therapy
- Patients with mental illness or substance use disorder
- Those using benzodiazepines
- People with other pain types, including visceral or neuropathic pain
- 87% of participants were male
Of 4,485 patients screened, only 265 enrolled
- 1,843 declined
- 2,377 excluded based on diagnosis, medication history, or comorbidities
Yet despite this narrow scope, the study is often used to justify restrictions on all chronic pain patients.
What Medications Were Used?
The opioid group received:
- Immediate-release morphine, oxycodone, or hydrocodone/acetaminophen
- Some were transitioned to extended-release meds or fentanyl patches
- Average daily dose: ~22 MME. This is well below therapeutic doses typically used for moderate-to-severe pain
The non-opioid group received:
- Acetaminophen, NSAIDs
- Adjuncts: duloxetine, gabapentin, nortriptyline
- Tramadol (an opioid) was used in up to 11% of patients by month 12
- The non-opioid group received more medications overall
The comparison was far from apples-to-apples — low-dose opioids were pitted against high-dose combinations of multiple meds, including another opioid.
What Were the Results?
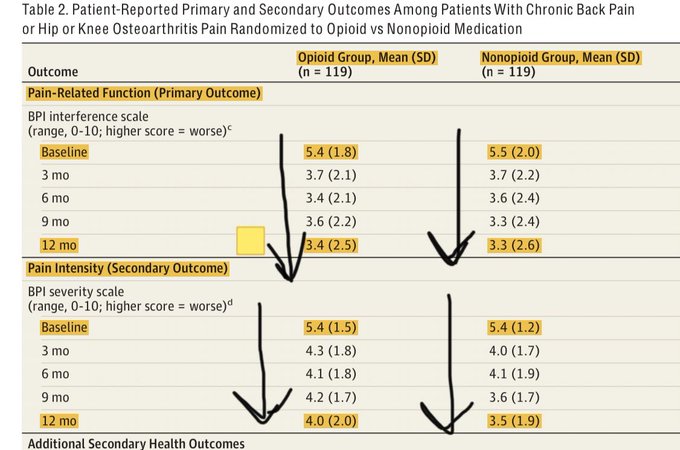
✅ Pain-Related Function (Primary Outcome)
Both groups improved
No significant difference at 12 months
-
Opioid group: 3.4
-
Non-opioid group: 3.3
-
(On 0–10 scale; higher = worse function)
-
Over 59% in each group had a ≥30% improvement in function
Pain Intensity (Secondary Outcome)
-
Slightly better in the non-opioid group
-
Opioid: 4.0
-
Non-opioid: 3.5
-
Statistically significant, but clinically small (~0.5)
-
Other Outcomes
- Anxiety scores improved more in the opioid group
- Quality of life scores were similar between groups
- Adverse effects (e.g., constipation, nausea) were more common in opioid group, but generally mild
- No deaths, no OUD, no doctor shopping, no diversion
🧠 What This Means
The SPACE Trial did not show that opioids are ineffective.
It showed that:
- Low-dose opioids and multi-drug non-opioid therapy had similar results
- Opioids were not more dangerous in terms of addiction, misuse, or death
- Anxiety was slightly better in the opioid group
- The differences in pain scores were small, and both groups benefited
This was a small, VA-based trial of opioid-naïve men with specific types of pain — not a referendum on treating all chronic pain conditions.
⚖️ The Litigation Narrative Begins Here
Despite its narrow scope and balanced findings, the SPACE Trial was immediately spun to support opioid lawsuits, with expert witnesses like Anna Lembke testifying under oath that:
“Researchers… were forced to conduct studies to counter the pharmaceutical companies’ opioid promotions. Those studies found that opioids were not effective for the long-term treatment of moderate to severe pain…”
But, the SPACE Trial showed opioids were effective — and that none of the 240 participants developed addiction. This is a continuation of a litigation-driven narrative that would later shape policy, funding, and widespread tapering — all built on a misrepresentation of the actual data.
📰 Section 3: How the Narrative Was Spun by Media and Advocacy Groups
Within days of its release, the SPACE Trial wasn’t just discussed — it was weaponized.
The findings, which showed that opioids and non-opioids performed similarly, were stripped of context and transformed into clickbait headlines and talking points for policy advocates, health agencies, and expert witnesses pushing for opioid elimination. The result? A misleading narrative that opioids “don’t work” — repeated so often it became accepted truth.
What the Headlines Claimed
Mainstream outlets ran with oversimplified — and in some cases, flat-out false — interpretations:
-
Vox: “Opioids are no better than other medications for some chronic pain.”
Source -
NPR: “Opioids are overrated for some common back pain, a study suggests.”
Source
🔍 These headlines implied that:
- Opioids don’t help at all
- They were clearly inferior
- They’re being overprescribed to people who don’t need them
None of that is supported by the actual trial data.
How PROP and Advocacy Groups Amplified It
Groups like PROP (Physicians for Responsible Opioid Prescribing) immediately used the study to support restrictive guidelines and lawsuit narratives.
In one PROP newsletter, they falsely claimed:
“The best evidence now shows that prescription opioids are ineffective for long-term management of common chronic pain conditions, such as osteoarthritis and low back pain.”
📌 But in the actual SPACE Trial:
- Both groups improved in pain and function
- No one developed addiction
- Function outcomes were equal
- Anxiety was better in the opioid group
Even duloxetine, gabapentin, and tramadol (an opioid) were used in the so-called “non-opioid” group — making the claim of opioid inferiority completely unsupported.
How This Became Legal & Policy Ammo
Policy influencers and expert witnesses — many of whom were also affiliated with PROP or paid by law firms — used these distorted claims to:
- Justify the CDC’s 2016 opioid guideline
- Support the VA and DoD “opioid-sparing” push
- Promote the idea that prescribing opioids equals harm
- Testify in opioid litigation that these findings prove opioids should not be used for chronic pain
This was the origin of the narrative we hear today:
“Opioids are ineffective. They're dangerous. They must be eliminated.” It didn’t come from the study itself. It came from those with something to gain — legally, politically, and financially.
💰Section 4: Who Benefited: Conflicts, Litigation, and Paid Experts
The misrepresentation of the SPACE Trial didn’t just happen in a vacuum. It happened because it served powerful interests — from expert witnesses in opioid lawsuits, to advocacy groups like PROP, to industries profiting from opioid elimination.
Litigation Strategy in Action
The SPACE Trial was cited repeatedly in opioid litigation, used to argue that opioids were ineffective and overprescribed. But it wasn’t just cited — it was directly shaped into a legal weapon.
Under oath in New York opioid litigation, Dr. Anna Lembke testified:
“Researchers, including those who published an article in the Journal of the American Medical Association in 2018, were forced to conduct studies to counter the pharmaceutical companies’ opioid promotions.”
She went on to claim:
“Those studies found that opioids were not effective for the long-term treatment of moderate to severe pain.”
🛑 Neither of those statements is accurate.
The SPACE Trial showed:
-
Opioids were effective for many patients
-
No one developed addiction or misuse
-
There was no overdose, diversion, or doctor-shopping
Lembke also testified in the same case that she earned $500–$800/hour as an expert witness, and admitted to earning hundreds of thousands of dollars doing so.
Judicial Pushback
Lembke’s claims have been challenged in court. In a 2022 California case, Judge Peter Wilson ruled that she:
“Overstated the risk of addiction”
And her testimony was “inconsistent with the evidence.”
Yet she continued to testify in other states, using the same flawed narratives — often based on the misrepresented SPACE Trial.
PROP, Guidelines, and Industry Ties
PROP (Physicians for Responsible Opioid Prescribing) also benefited from the study’s spin:
- PROP’s members helped write the 2016 CDC Guidelines
- They frequently referenced the SPACE Trial as evidence that opioids don’t work
- Several PROP-affiliated experts served as paid litigation witnesses in opioid lawsuits
In one newsletter, PROP claimed:
“The best evidence now shows that prescription opioids are ineffective for long-term management of common chronic pain conditions.”
This directly contradicts the actual study, where:
- Both groups improved
- Function was the same
- No OUD cases were detected
Abatement Money: The More Diagnoses, the More Dollars
In opioid lawsuits, settlement abatement funds were distributed based on the number of people diagnosed with opioid use disorder (OUD). The more people labeled with OUD, the more money states and counties received.
This created a perverse incentive:
- Misclassify pain patients as having OUD
- Use inflated misuse stats to secure funding
- Justify denying opioids and promoting “treatment” (often Suboxone) even in cases where addiction wasn’t present
The SPACE Trial, when spun to suggest opioids “don’t work,” became part of that narrative — even though the actual study found no OUD or misuse.
Who Gained
- Litigators made billions in settlements
- Addiction pharma companies gained market dominance (e.g., Suboxone)
- Expert witnesses were paid hundreds of thousands
- PROP-aligned policymakers pushed restrictive guidelines
- “Anti-opioid” groups got grants, funding, and influence
Who lost? Chronic pain patients — cut off, tapered, misdiagnosed, and forgotten
🩹Section 5: How it Was Misused to Harm Patients
The distorted narrative around the SPACE Trial didn’t just stay in courtrooms or academic journals. It was used to justify sweeping, dangerous policies that cut off care to millions of pain patients — many of whom were never even part of the population studied in the trial.
Misapplied to the Wrong Patients
Although the SPACE Trial:
- Included only opioid-naïve veterans
- Focused only on back and OA pain
- Excluded people with fibromyalgia, migraines, mental illness, or prior opioid use
- Used low-dose opioids (~22 MME/day)
Its conclusions were broadly applied to:
- Patients on high-dose, long-term opioid therapy
- Women with endometriosis, lupus, CRPS
- People with sickle cell disease, cancer survivorship pain, and rare disorders
- Patients already dependent on opioids for basic function
Policies were enacted as if this trial proved that opioids don’t work for anyone. They didn’t — and it didn’t.
Used to Justify Forced Tapering
The SPACE Trial became a go-to citation for:
- Insurance companies denying coverage for opioid therapy
- Doctors tapering patients against their will
- Hospitals cutting off post-surgical opioids
- Regulators punishing physicians who prescribed above “low-dose” levels
- Never mind that the trial:
- Found no cases of OUD or misuse
- Reported better anxiety scores in the opioid group
- Involved none of the patients now being tapered or abandoned
What Was Actually Found?
Let’s be clear about what the study showed:

Yet somehow, this was twisted into “Opioids don’t work” and “They’re too dangerous to prescribe.”
Quotes That Prove the Misuse
PROP Newsletter:
“The best evidence now shows that prescription opioids are ineffective for long-term management of common chronic pain conditions…”
Anna Lembke (interview):
“If opioids worked long-term, I would have no problem with patients taking them… [but] they stop working and then you have an additional problem.”
But her own citation — the SPACE Trial — showed they do work for many, even at low doses, and without resulting in addiction.
Fallout: The Wrong Takeaways, The Wrong Target
Instead of learning that:
- Multiple treatments can help
- Low-dose opioids may be effective and safe
- Patients should have individualized options
Officials took away:
- “Opioids don’t help anyone”
- “High doses are unjustified”
- “We need to restrict access and punish providers”
This has led to:
- Thousands of forced tapers
- Countless ER visits for untreated pain
- Increased suicides and suffering
- Patients being labeled with OUD who were never misusing
All based on a study that never showed what they say it did.
🎯Section 6: What the Study Really Means
The SPACE Trial was never a sweeping judgment on opioids — it was a limited, controlled comparison of two treatment strategies in a small, specific group of patients. And when you strip away the headlines, soundbites, and litigation spin, here’s what it actually tells us:
Opioids Can Work — Even at Low Doses
Despite the study using low-dose opioids (~22 MME/day), many patients saw improvement:
- Pain intensity dropped
- Function improved
- Anxiety was reduced
Over 59% in both groups had a ≥30% improvement in pain-related function — including the opioid group, even with a modest dose.
It Doesn’t Justify Forced Tapering or Denial of Care
The trial did not:
- Study people with complex, long-term chronic pain
- Include anyone already on opioids
- Test what happens when opioids are taken away
- Show harm from long-term opioid therapy
Yet it's being used to justify cutting off care for patients it didn’t even study.
It Reported Zero Cases of Addiction, Misuse, or Overdose
Out of 240 patients, there were:
- 0 cases of OUD
- 0 cases of doctor-shopping or diversion
- 0 overdose deaths
Even PROP and addiction medicine advocates can’t point to this study as evidence of harm — because there was none.
It Was Misused to Support a Litigation Narrative
The SPACE Trial was used as “evidence” that opioids are ineffective — but only after being spun by advocacy groups, media, and expert witnesses who stood to benefit financially or politically. This wasn’t science speaking. It was strategy.
The Authors Themselves Admitted Its Limits
Even Dr. Erin Krebs, the lead author, acknowledged:
“It’s a bit too early for our study findings to be incorporated into guidelines.”
She also admitted that critics of the study were dismissed as "industry-funded," rather than actually addressing concerns about misapplication.
Final Takeaway
This trial was never meant to be the final word on opioid therapy.
But it was taken that way — and used to create a policy environment where:
- Pain patients were cut off
- Doctors were punished
- Opioid therapy was demonized
- And truth was buried under spin
It’s time to stop misusing this study — and start rebuilding pain care on evidence, not ideology.
Free Printable Resources: Please subscribe (free) to our Patreon Page for:
- Printable Summary
- Q&A for patients and advocates
- Myths v Facts